|
|
|
|
This family of nonenzymatic polypeptides contain 60-74 amino acid residues and four or five disulfide bridges. Four disulfide bridges are conserved in all the members. Consequently, they share similar pattern of protein folding: three β-stranded loops extending from a central core containing the four conserved disulfide bridges. Because of their appearance, this family of proteins is called the three-finger toxin family. However, they exhibit distinct pharmacological properties including neurotoxic, cardiotoxic, anticoagulant and antiplatelet effects. Therefore their structure-function relationships are subtle and complicated. We solved structure-function relationships of this family of proteins using theoretical methods. The following are some of our contributions to the structure-function relationships of three-finger toxins
- Cytolytic site – Using hydropathic profiles and charge distribution, we showed that myotoxic region in phospholipase A2 enzymes consists of a hydrophobic and cationic segments next to each other (Phospholipase A2 enzymes). We extended these studies other cytolytic proteins and found that they all share the similar characteristics (Common cytolytic region). Unlike other cytolysins, the cationic and hydrophobic sites are formed as a result of their folding in cardiotoxins/cytotoxins (a subgroup of three-finger toxins). Thus they have conformational cytolytic sites. We have studie
 d the importance of this cationic site by chemical modification of Lys residues in cardiotoxins. When the positive charges of the Lys residues are changed to negative or neutral charges by succinylation or carbamylation, respectively, cardiotoxin loses its cytolytic activity, but when guanidinated and the positive charges are retained, the cytolytic activity remains intact. A body of compelling experimental evidence has accumulated over the years to support this common cytolytic region in other cytolytic proteins. d the importance of this cationic site by chemical modification of Lys residues in cardiotoxins. When the positive charges of the Lys residues are changed to negative or neutral charges by succinylation or carbamylation, respectively, cardiotoxin loses its cytolytic activity, but when guanidinated and the positive charges are retained, the cytolytic activity remains intact. A body of compelling experimental evidence has accumulated over the years to support this common cytolytic region in other cytolytic proteins.
- Hypotensive site – Calciseptine is a potent blocker
 of L-type calcium channel isolated from mamba snake venom. By systematic comparison of the sequences of three-finger toxins from mamba venoms and using proline brackets theory (Protein - protein interaction sites), we identified the functional site of this and other L-type calcium c of L-type calcium channel isolated from mamba snake venom. By systematic comparison of the sequences of three-finger toxins from mamba venoms and using proline brackets theory (Protein - protein interaction sites), we identified the functional site of this and other L-type calcium c hannel blockers. The synthetic peptide, L-calchin, shows negative inotropic effects in isolated rat hearts similar to parent protein. It also blocks the L-type calcium current in rabbit cardiac myocytes as shown by patch clamp experiments. This block is voltage-independent. Thus, the 8-residue peptide behaves similar to the parent molecule. hannel blockers. The synthetic peptide, L-calchin, shows negative inotropic effects in isolated rat hearts similar to parent protein. It also blocks the L-type calcium current in rabbit cardiac myocytes as shown by patch clamp experiments. This block is voltage-independent. Thus, the 8-residue peptide behaves similar to the parent molecule.
- Analgesic site – Hannalgesin from King cobra venom shows potent anal
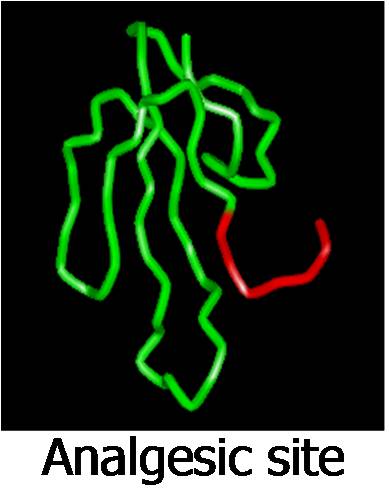 gesic effects. In addition, this protein is a potent neurotoxin. Using proline brackets theory (Protein-protein interaction sites), we identified the functional site of hannalgesin. The short peptide of 11 amino acid residues exhibits potent analgesic effects without any neurotoxicity. The peptide is active by sublingual, oral and intraperitoneal routes in mice and rats. gesic effects. In addition, this protein is a potent neurotoxin. Using proline brackets theory (Protein-protein interaction sites), we identified the functional site of hannalgesin. The short peptide of 11 amino acid residues exhibits potent analgesic effects without any neurotoxicity. The peptide is active by sublingual, oral and intraperitoneal routes in mice and rats.
|
Key
Publications
|
|

